|
Which properties can be identified using periodic trends? Electronegativity:Įlectronegativity is a property that measures the tendency of an atom to attract electrons to form a bond. We discuss trends for properties such as electronegativity, atomic radius, first ionization energy, reactivity and electron affinity. This is due to the structural similarities’ elements have within a period or family that allows for these trends to take place. These trends have allowed scientists in the past to predict certain characteristics of unknown elements. To see how well you know the information, try the Quiz or Test activity.Periodic trends are observable patterns in the properties of an element that are dependent on its position in the Periodic Table. When you need a break, try one of the other activities listed below the flashcards like Matching, Snowman, or Hungry Bug.Īlthough it may feel like you're playing a game, your brain is still making more connections with the information to help you out. 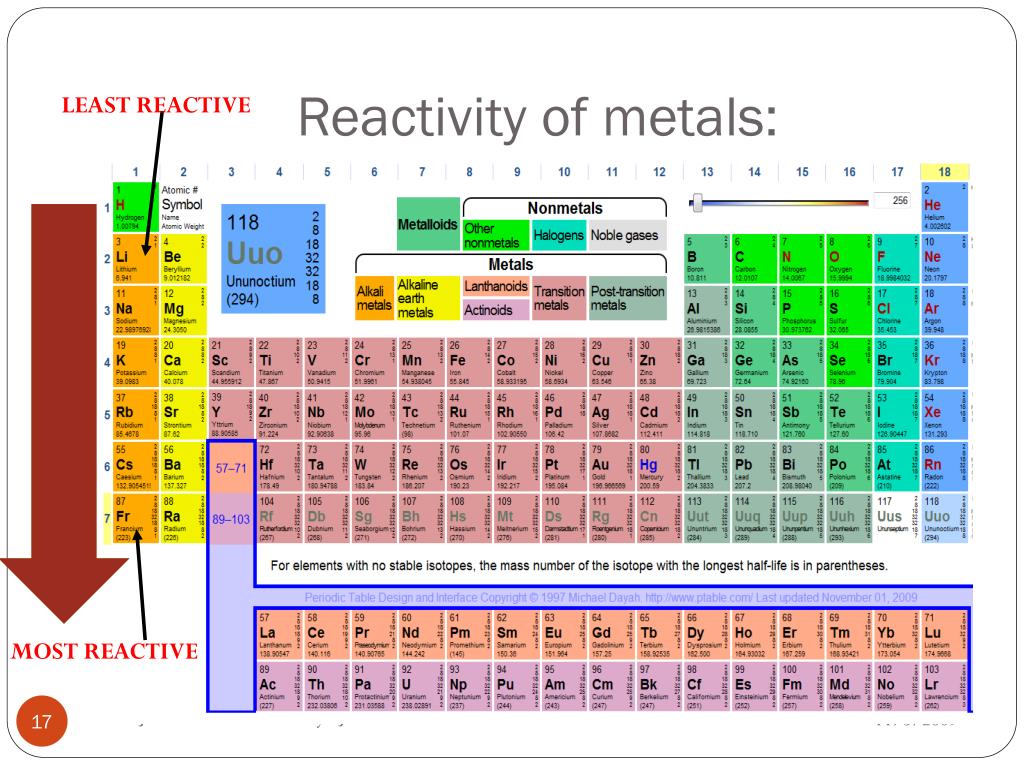
If you are logged in to your account, this website will remember which cards you know and don't know so that theyĪre in the same box the next time you log in. 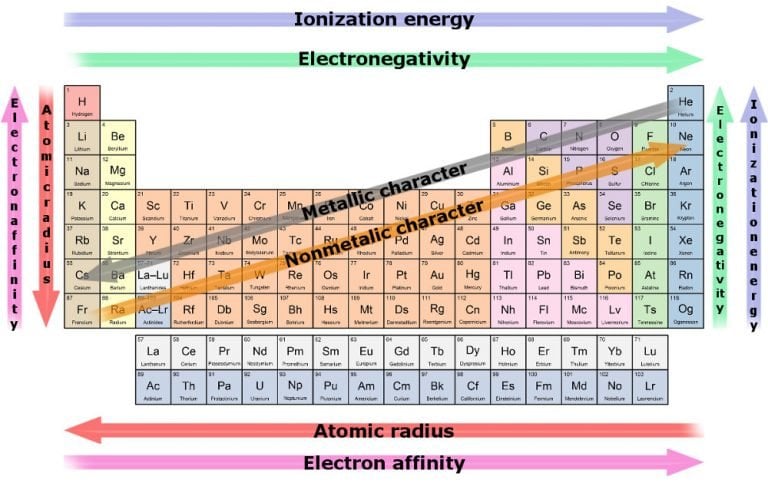
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
